Classroom Chemistry
Chemistry is the study of how substances and materials interact with one another.
Matter is every thing that takes up space and has weight!
States of Matter Game!!!
Here is a cool video to show us changes and states of matter.
Here is one to help us understand properties of matter!
Here is one to help us understand properties of matter!
Properties of Matter Game
Mixtures and Solutions!!!
Solute- a substance that can be dissolved from its solid form into a liquid.
Solvent- a substance that holds the dissolved solute.
Solutions are liquid mixtures that have a solute dissolved in the solvent.
They hard to separate because their physical properties change when mixed!
Usually the only way to separate them is through evaporation.
Mixtures are easily separated because they keep their physical properties!
There are 3 different types of mixtures:
Homogeneous, Heterogeneous, and Colloid
Homogeneous mixtures - look like they are one substance (salt and sugar)
Heterogeneous mixtures - are substances that do not mix together to look like one substance (sand and cheerios)
Colloid mixtures look like homogeneous mixtures, but actually are not! The particles inside the mixture are so small that you can not see that they are just evenly distributed in the mixture! (milk in water)
Solvent- a substance that holds the dissolved solute.
Solutions are liquid mixtures that have a solute dissolved in the solvent.
They hard to separate because their physical properties change when mixed!
Usually the only way to separate them is through evaporation.
Mixtures are easily separated because they keep their physical properties!
There are 3 different types of mixtures:
Homogeneous, Heterogeneous, and Colloid
Homogeneous mixtures - look like they are one substance (salt and sugar)
Heterogeneous mixtures - are substances that do not mix together to look like one substance (sand and cheerios)
Colloid mixtures look like homogeneous mixtures, but actually are not! The particles inside the mixture are so small that you can not see that they are just evenly distributed in the mixture! (milk in water)
Tools to Separate!
Some tools we use to separate mixtures and solutions include: Magnets, Strainer, Water, Filter, Hot Plate, Beaker, and Tweezers.
Water is the universal solvent (liquid) and the solute is what gets dissolved into the solvent.
Some tools we use to separate mixtures and solutions include: Magnets, Strainer, Water, Filter, Hot Plate, Beaker, and Tweezers.
Water is the universal solvent (liquid) and the solute is what gets dissolved into the solvent.
Lab 1 - Mixtures: Liquid and Solids
Lab 2: Mixtures and Solutions: Liquids and Liquids
Lab 3: Separating Mixtures
Making Geodes by Growing Crystals!
In this lab we will learn how crystals grow!
Crystals are formed from vapors, solutions, or melts! Pressure, heat, or evaporation causes a chemical reaction and some of the atoms and molecules are forced to change and join into a cluster. This is the start of a crystal and as more molecules and atoms join together, they grow bigger and bigger!
We will be creating our geodes by making a saturated alum powder solution and setting an egg shell will dried glue and alum powder on it, into a solution. As we continue to saturate the solution and add it back to the crystals, they will get bigger!!! Don't be afraid to try this at home after we have done it!!!
In this lab we will learn how crystals grow!
Crystals are formed from vapors, solutions, or melts! Pressure, heat, or evaporation causes a chemical reaction and some of the atoms and molecules are forced to change and join into a cluster. This is the start of a crystal and as more molecules and atoms join together, they grow bigger and bigger!
We will be creating our geodes by making a saturated alum powder solution and setting an egg shell will dried glue and alum powder on it, into a solution. As we continue to saturate the solution and add it back to the crystals, they will get bigger!!! Don't be afraid to try this at home after we have done it!!!

Surface Tension!
Surface tension is the film that forms on the surface of a liquid caused by cohesion. Cohesion is the force of attraction between particles (molecules) of a substance. We predicted that a penny wouldn't hold that many drops of water on its surface, but we were surprised to find out that it actually hold more than we thought!!! When we added soap to the water we also discovered that it decreased the cohesion of the water molecules which then decreased the surface tension, so we could put half as many drops of water on the penny!
Surface tension is the film that forms on the surface of a liquid caused by cohesion. Cohesion is the force of attraction between particles (molecules) of a substance. We predicted that a penny wouldn't hold that many drops of water on its surface, but we were surprised to find out that it actually hold more than we thought!!! When we added soap to the water we also discovered that it decreased the cohesion of the water molecules which then decreased the surface tension, so we could put half as many drops of water on the penny!
Physical and Chemical Changes!
|
|
|
|
Found at TPT: Evidence of Chemical Changes
|
Found at TPT: Evidence of Physical Changes
|
Evidence of Chemical Reactions where a Gas is Produced.
In this lab we saw how much gas is produced when we added baking soda to vinegar by placing a balloon on top! It was very cool to see it fizz up all the way into the balloon! We also noticed a huge temperature change which is also evidence of a chemical reaction occurring!
Baking soda and vinegar react with each other because of an acid-base reaction. Baking soda is a bicarbonate (base) (NaHCO3) and vinegar is an acetic acid (HCH3COO). One of the products this reaction creates is carbon dioxide.
How is the air we breathe different from CO2?
We breathe oxygen (O2). CO2 has a carbon molecule attached to the oxygen. Our body does not use the carbon. In fact, our body absorbs the oxygen and when we are done with it, in our bodies, we breathe it out. When we breathe it out we also breathe out the carbon in our bodies too.
Baking soda and vinegar react with each other because of an acid-base reaction. Baking soda is a bicarbonate (base) (NaHCO3) and vinegar is an acetic acid (HCH3COO). One of the products this reaction creates is carbon dioxide.
How is the air we breathe different from CO2?
We breathe oxygen (O2). CO2 has a carbon molecule attached to the oxygen. Our body does not use the carbon. In fact, our body absorbs the oxygen and when we are done with it, in our bodies, we breathe it out. When we breathe it out we also breathe out the carbon in our bodies too.
Formation of Carbon Dioxide Video
and Game
Reversible or Irreversible
Some changes in chemistry are reversible, while others are not.
Reversible Examples: Turning water into Ice
Irreversible Examples: Burning paper
Reversible Examples: Turning water into Ice
Irreversible Examples: Burning paper
Reversible or Irreversible Game
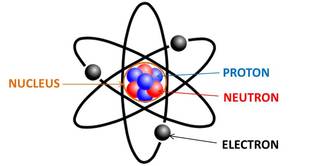
Atoms
Everything in the world is made up of atoms. The picture below is a diagram of what an atom is made from. Atoms are made up of different molecules such as a nucleus (the brain), protons (positively charged), electrons (negatively charged), and neutrons (neutral / no charge).
Everything in the world is made up of atoms. The picture below is a diagram of what an atom is made from. Atoms are made up of different molecules such as a nucleus (the brain), protons (positively charged), electrons (negatively charged), and neutrons (neutral / no charge).
Hydrogen is a chemical element with the symbol H. It is atomic number 1 and the lightest element on the periodic table. A hydrogen ion is created when the nucleus of a hydrogen atom is separated from its accompanying electron. The hydrogen nucleus is made up of a proton. The single hydrogen ion, represented by the symbol H +, is therefore customarily used to represent a proton. Wikipedia
AcidsAn acid is a substance that donates hydrogen ions. Because of this, when an acid is dissolved in water, the balance between hydrogen ions and hydroxide ions is shifted. Now there are more hydrogen ions than hydroxide ions in the solution. This kind of solution is acidic.
|
BasesA base is a substance that accepts hydrogen ions. When a base is dissolved in water, the balance between hydrogen ions and hydroxide ions shifts the opposite way. Because the base "soaks up" hydrogen ions, the result is a solution with more hydroxide ions than hydrogen ions. This kind of solution is alkaline.
|
Indicator Strips Lab
Click here if you would like to study some of our vocab words using flash cards!!!
Homework!!!!!
You are required to fill in the answers for the study guide using the website and our notes from our duo-tangs!
We will be learning how to:
1. Recognize and identify examples of mixtures of two or more solids, a solid and a liquid, and two or more liquids
2. Applying and evaluate a variety of techniques for separating different materials and mixtures.
3. Distinguishing substances that will dissolve in a liquid from those that will not, and demonstrate a way of recovering a material from solution.
4. Making a crystal!
5. Recognizing that the surface of water has distinctive properties
6. Describe the interaction of water with liquids and solids.
7. Produce carbon dioxide gas through the interaction of solids and liquids, and demonstrate how it's different from air
8. Distinguish reversible from irreversible changes of materials, and give examples of each
9. Recognize and describe evidence of a chemical reaction.
10. Explain how the products of a reaction differ from the original substances.
11. Use an indicator to identify a solution as being acidic or basic.
We will also review wearing safety equipment!!!
1. Recognize and identify examples of mixtures of two or more solids, a solid and a liquid, and two or more liquids
2. Applying and evaluate a variety of techniques for separating different materials and mixtures.
3. Distinguishing substances that will dissolve in a liquid from those that will not, and demonstrate a way of recovering a material from solution.
4. Making a crystal!
5. Recognizing that the surface of water has distinctive properties
6. Describe the interaction of water with liquids and solids.
7. Produce carbon dioxide gas through the interaction of solids and liquids, and demonstrate how it's different from air
8. Distinguish reversible from irreversible changes of materials, and give examples of each
9. Recognize and describe evidence of a chemical reaction.
10. Explain how the products of a reaction differ from the original substances.
11. Use an indicator to identify a solution as being acidic or basic.
We will also review wearing safety equipment!!!